Keys to Co-op
In-Practice Branding
Create awareness and education of NeuroStar for patients inside
your practice so they ask you about NeuroStar.
Create awareness and education of NeuroStar for patients inside
your practice so they ask you about NeuroStar.
Patients who have struggled to find help for their depression often just need to know there’s another option, and to be given the opportunity to ask for it. With our in-practice co-branded assets, and NeuroStar clothing and accessories, patients will become aware you offer NeuroStar and ask you about it.
Use co-op dollars to cover up to 50% of:
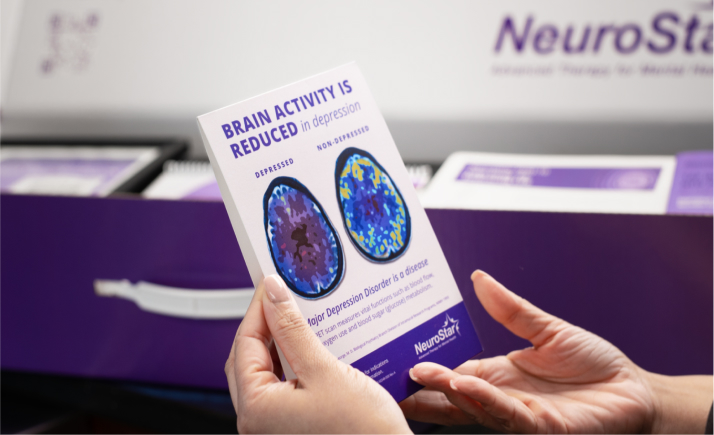
Successful In-Practice branding is more than just getting the right materials – it’s using them in the right ways. We’ve created the “3-1-1 System” for practice branding success.
3-1-1 means:

These pieces of collateral can be stand-up banners, printed brochures and information cards.

Copyright @ 2025 Neuronetics. All Rights Reserved.
Adult Indications for Use
The NeuroStar Advanced Therapy System is indicated for the treatment of depressive episodes and for decreasing anxiety symptoms for those who may exhibit comorbid anxiety symptoms in adult patients suffering from Major Depressive Disorder (MDD) and who failed to achieve satisfactory improvement from previous antidepressant medication treatment in the current episode.
The NeuroStar Advanced Therapy System is intended to be used as an adjunct for the treatment of adult patients suffering from Obsessive-Compulsive Disorder (OCD).
Adolescent Indications for Use
NeuroStar Advanced Therapy is indicated as an adjunct for the treatment of Major Depressive Disorder (MDD) in adolescent patients (15-21).
NeuroStar Advanced Therapy is only available by prescription. A doctor can help decide if NeuroStar Advanced Therapy is right for you. Patients’ results may vary.
Important Safety Information
The most common side effect is pain or discomfort at or near the treatment site. These events are transient; they occur during the TMS treatment course and do not occur for most patients after the first week of treatment. There is a rare risk of seizure associated with the use of TMS therapy (<0.1% per patient).
NeuroStar Advanced Therapy should not be used with patients who have non-removable conductive metal in or near the head. NeuroStar Advanced Therapy has not been studied in patients who have not received prior antidepressant treatment.

Copyright @ 2021 Neuronetics. All Rights Reserved.
Visit https://neurostar.com for indications for use and safety information
| Cookie | Duration | Description |
|---|---|---|
| cookielawinfo-checkbox-analytics | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Analytics". |
| cookielawinfo-checkbox-functional | 11 months | The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". |
| cookielawinfo-checkbox-necessary | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". |
| cookielawinfo-checkbox-others | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. |
| cookielawinfo-checkbox-performance | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Performance". |
| viewed_cookie_policy | 11 months | The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. It does not store any personal data. |